Statement from FDA Commissioner on Insulin Pricing
The FDA has issued a statement intended to spur competition and lower prices. I’ve pulled out and included a few passages that are about insulin. You can read the entire statement as written here.
from FDA.gov
Statement from FDA Commissioner Scott Gottlieb, M.D., on new actions advancing the agency’s biosimilars policy framework.
“There are currently no approved insulin products that can be substituted at the pharmacy level. One reason is that it was hard to bring a substitutable generic insulin to the market under the conventional drug pathway. The biosimilar pathway should make this kind of competition more accessible. Once an interchangeable insulin product is approved and available on the market, it can then be substituted for the reference product at the pharmacy, potentially leading to increased access and significantly lower costs for patients.”
We’re going to be monitoring these markets. And we’ll be taking additional actions. We’re actively evaluating how we can make it easier for biosimilar manufacturers to use reference products from outside the U.S., where prices may be cheaper and reference products more accessible.
We’re also releasing today two critical guidance documents that describe how the agency plans to implement Congress’ direction that we transition certain biological products currently approved as drugs under the FD&C Act to be licensed as biologics under the PHSA.
Transitioning these drugs to the PHSA will let them to be treated as biologics under that law. And that means opening them up to competition through the biosimilars pathway. This includes insulin, which has been historically regulated as a drug and not a biologic.
Starting in March 2020, the approved marketing applications for the small subset of “biological products” such as insulin and human growth hormone – which for complex historical reasons were previously generally approved as drugs under section 505 of the FD&C Act – will be deemed to be biologics licenses under section 351 of the PHSA. Sponsors have known about this transition for a decade. They’ve had time to prepare.
“As a result, we’ve heard frequent reports of patients rationing insulin, and in some cases dying because they can’t afford the injections they need to survive. These tragic stories aren’t isolated occurrences. And they’re not acceptable for a drug that’s nearly a century old.”
Today, we’re laying out our policy on how these products will transition from the drug pathway to the biologics pathway, and in so doing, how we intend to use this new framework to promote competition. The two guidance documents we’re releasing today, one final and one draft, describe how the FDA intends to accomplish the transition of these products under the “Deemed to be a License” provision of the BPCI Act. The final guidance deals with “Interpretation of the ‘Deemed to be a License’ Provision of the Biologics Price Competition and Innovation Act of 2009,” finalizes the FDA’s draft guidance from 2016.
We’ve shaped these policies to implement the intent of Congress, and to make sure a few things happen. First, that the anti-evergreening provisions under the biosimilars legislation – meant to prevent sponsors from being able to game the exclusivity provisions to forestall biosimilar entry – will apply to these newly deemed products, including insulin.
Breaking News! The FDA decision on Dexcom G5 Dosing Is In!
Thanks to Bennet and our friends at DPAC for the up-to-the minute information!
from Diabetes Patient Advocacy Coalition
FDA's Advisory Committee meeting just voted to recommend approval of the expansion of the Dexcom G5 CGM label.
The results are in and the FDA panel has voted the following for the labeling change of the Dexcom G5 Continuous Glucose Monitoring System:
1. Is there reasonable assurance that the Dexcom G5 Continuous Glucose Monitoring System is safe for the proposed indications of use? VOTE - YES: 8 NO: 2
2. Is there reasonable assurance that the Dexcom G5 Continuous Glucose Monitoring System is effective for the proposed indications for use? VOTE - YES: 9 NO: 1
3. Do the benefits of the Dexcom G5 Continuous Glucose Monitoring System for the proposed indications of use outweigh the risks of the Dexcom G5 Continuous Glucose Monitoring System for the proposed indications for use. VOTE - YES: 8 NO: 2
You Could Be a Consumer Representative on an FDA Advisory Committee
from fda.gov
By: CAPT Dornette Spell-LeSane, M.S.N., M.H.A., A.N.P.-B.C.
Have you ever wanted to be part of the food and drug regulatory process? Do you have a history of public interest or a passion for consumer advocacy? Do you have experience analyzing scientific data?
CAPT Dornette Spell-LeSane, M.S.N., M.H.A., A.N.P.-B.C.
[Dornette Spell-LeSane] If you answered “Yes,” here’s your opportunity to become an advocate for consumers! The Food and Drug Administration continually seeks input from consumers on scientific and medical issues by including Consumer Representatives on Agency advisory committees.
Participation as a Consumer Representative requires a modest time commitment. Travel expenses are paid and representatives receive reasonable compensation.
Consumer Representatives serve as Special Government Employees on a committee for up to a four-year term. Committees meet 1-3 times annually for 1-2 days. All meetings are held in the Washington, D.C. area. Members receive per-diem and travel expenses and are paid at a GS-15/10 hourly rate for the days attending a meeting.
Consumer Representatives provide the perspective of consumers to advisory committees and do not represent their own personal expertise. Their role is to:
Represent the consumer perspective on issues and actions before the advisory committee;
Serve as a liaison between the committee and interested consumers, associations, coalitions, and consumer organizations; and, Facilitate dialogue with the advisory committees on scientific issues that affect consumers.
if you want to learn more including how to apply... click here.
Dexcom G4 Platinum Continuous Glucose Monitoring System WITH Share
Dexcom receives FDA approval for their G4 Platinum Continuous Glucose Monitoring System WITH Share, "faster than expected" and that's great news for tons of reason.
On Monday January 26, 2015 Dexcom announced in a press release that their G4 Platinum Receiver with Share built in was approved by the FDA in under 120 days (Thank you FDA!). Later in the day Dexcom held a webcast to make the announcement. I've listened to the webcast, pulled out the most interesting parts and included them for you here in my third and final microphone test before my new podcast officially launches.
If you listened to the audio you just heard that there is some crazy exciting news coming out of Dexcom's headquarters this week that's going to benefit everyone who uses a Dexcom CGM.
If you weren't able to listen here are just a few details from the announcement plus links to Dexcom's complete press release and webcast.
- The Share receiver is anticipated to ship to new patients in early March 2015.
- All patients who purchased a Share cradle will receive a free upgrade to the Share receiver.
- All purchasers of a G4 Platinum receiver from January 1, 2015, until the Share receiver is shipped, will receive a free upgrade to the Share receiver.
- There will be a low cost cash upgrade to the Share receiver for those patients who are still under warranty with their existing receiver.
- The Share receiver will be compatible with future generation Dexcom sensor systems.
The entire press release is here
The entire webcast is here
Dexcom's New 505 Software Review and How To
The (Algo) Rhythm is Gonna Get You!
Last week the good folks at the FDA approved Dexcom's latest software for use in your G4 continues glucose monitor. Let's talk about what you'll need to know about installing the software on your receiver and if I saw a difference after upgrading.
What you need to upgrade?
A Dexcom G4 adult receiver (The company has announced that they will submit the software to the FDA in 2014 for approval in their pediatric receiver).
A Windows computer with Internet access
The charging cable from your Dex, minus the electric adapter, so USB to Dexcom
A few minutes
What to do...
Go to Dexcom's website and click on the "Download NEW Software 505" link. You will need to login to the Dexcom site, if you do not have an online account on their site, you can quickly make one.
A downloader program is installed on your computer. After launch it will ask for you to connect your receiver to the computer and the rest happens in short order. Please note TWO important things. The process will revert your receiver back to it's factory specs. Make notes of all your settings and be prepared to reconfigure the receiver (takes a few minutes) after installation. Also, you will have to restart your sensor session after the update, so you won't be getting any of that good BG data during those two hours - plan accordingly.
You don't have to wait until it's time to put on a new sensor to upgrade! Just choose 'Stop Sensor' on your receiver's menu and then restart the receiver after the software upgrade has finished. You DO NOT have to change your sensor site to do a sensor stop/start.
Should I do it?
Yes, here's why...
Dexcom is reporting that the MARD (mean absolute relative difference) will increase by 4%. In plain language, your Platinum G4 without the new algorithm has a 13% MARD , the new software clocks in at 9% MARD - a lower MARD is more accurate.
Did you know? The new 505 software is the version currently being used in artificial pancreas trials!
What did I see after the upgrade?
We upgraded Arden's receiver and have been running it for a week in conjunction with another G4 receiver that is using the previous software. My in a nutshell review -- the new software has been consistently closer to our finger stick checks than the previous software. We use the OmniPod meter with Freestyle strips. This is not to say that the previous software is always farther off then that of the newer version - at times it has been and at times it hasn't. The newer version (505) has been consistent with my prior expectations or better, never worse. But when it is appreciably better, it is 20 - 30 points more accurate and that's worth upgrading for in my opinion. It also seems to deliver more accurate fall and rise rates that don't linger after the change in BG has leveled.
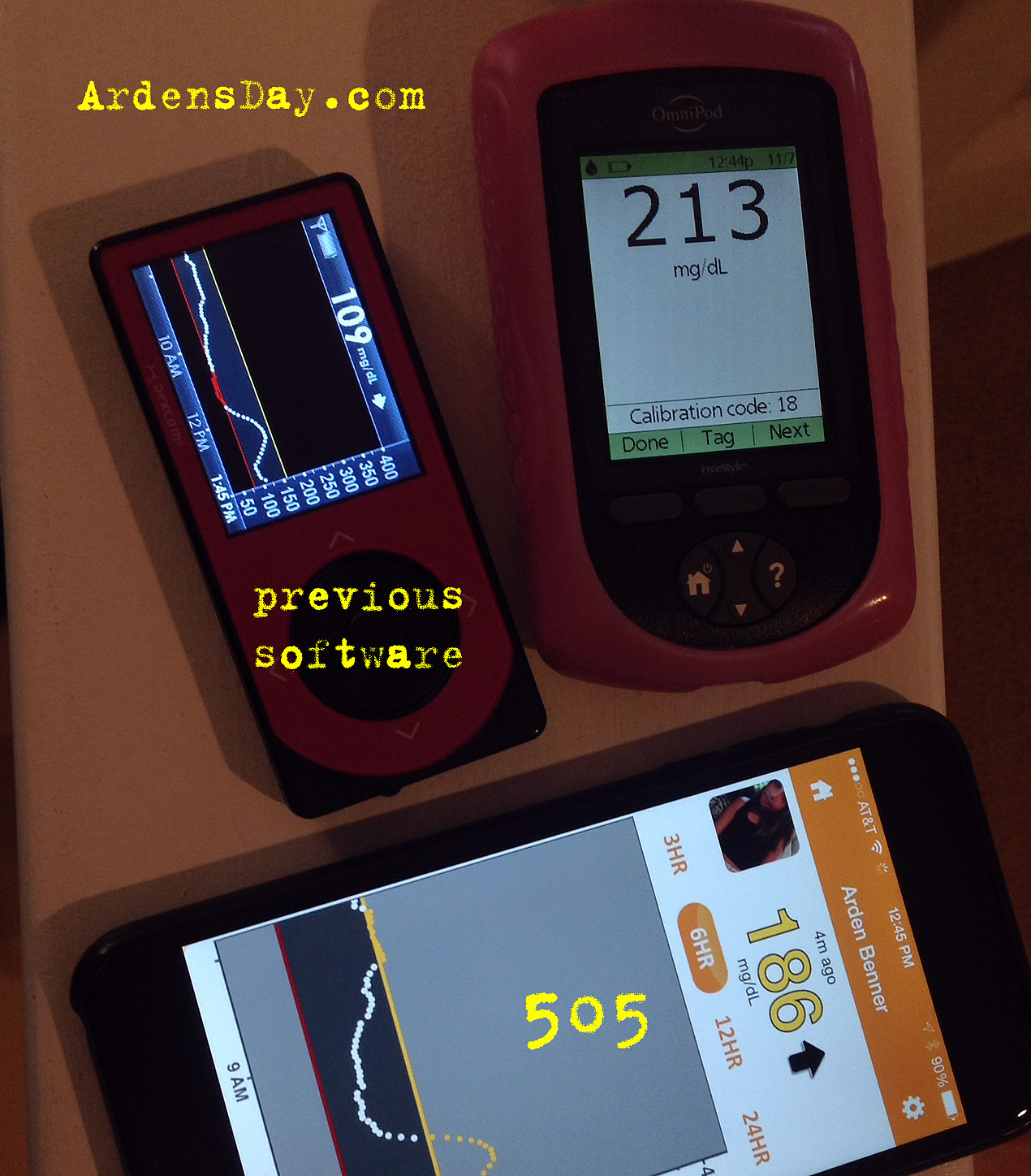
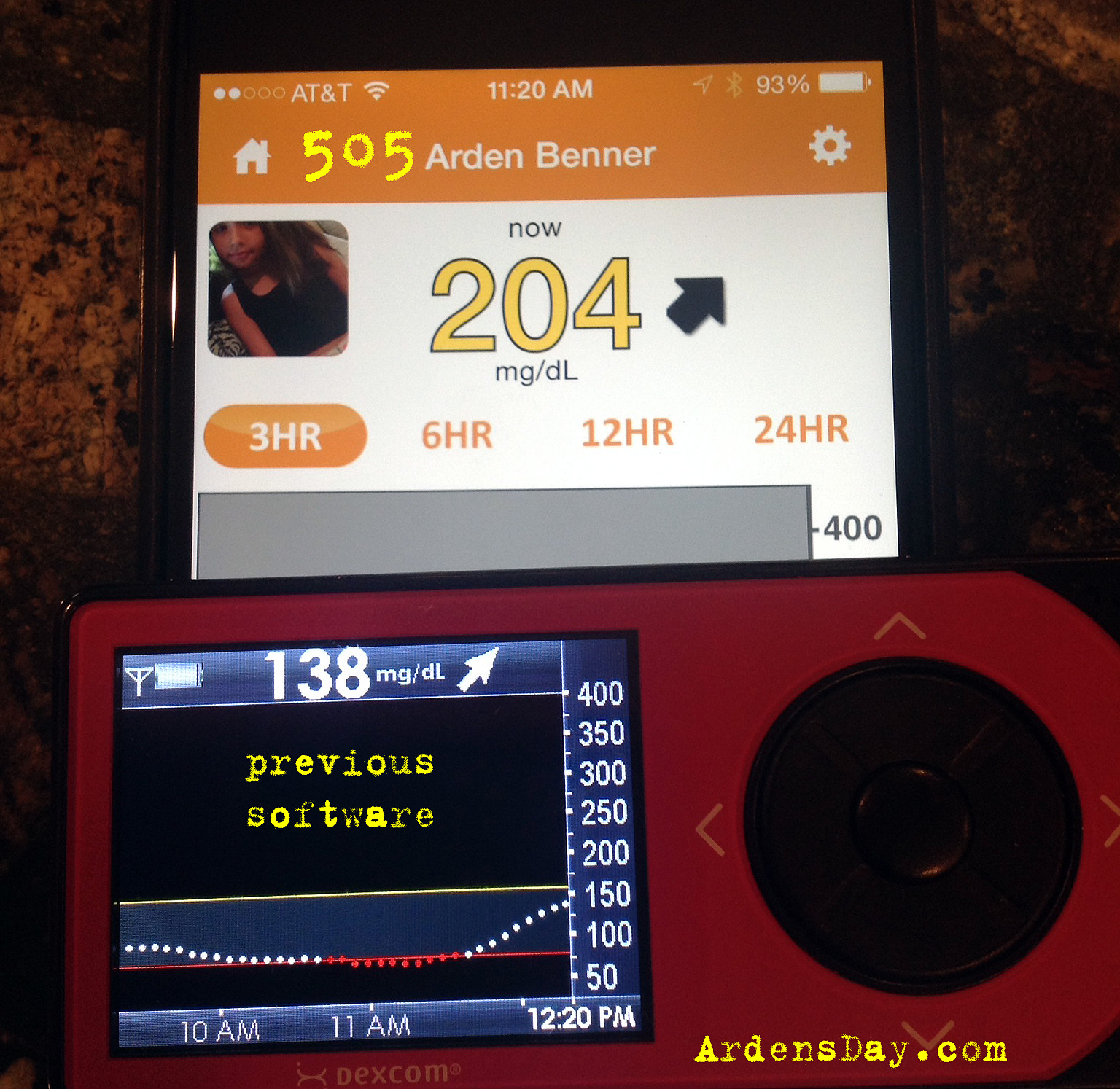
Here's some side-by-side pics...
The first four images show a BG rise (I confirmed with a meter not pictured). You can see that the previous software lagged behind the rise and never reached the actual BG which topped out at 213 (on meter). The fifth image is an example of no significant difference between 505, previous and meter. The last image shows a metered 68 that registered as a 6o with the new software but an alarming 46 with the previous version. After testing and calibration the previous version adjusted to 60, the new version adjusted to 66.
Conclusion...
Obviously my observations are just that, observations. Nothing very scientific was done and I am not a doctor as my disclaimer mentions. The upgrade is fast and simple but Mac users will have to bum a Windows machine from a friend.
The 505 software is currently not approved for the pediatric version of the G4 but Dexcom announced that they will be applying with the FDA in the last quarter of 2014 - that's any day.
The upgrade is free, better and the next step toward CGM data that perhaps one day you'll be able to dose insulin from - might as well do it... I don't see any downside.
Managing type 1 Diabetes just got a little bit easier in my opinion and I expect to see a difference in Arden's A1c after a full three months with the new software - time will tell.